Some people confuse the words nitrite and nitrate, but they are different compounds. Both nitrites and nitrates are naturally occurring inorganic compounds composed of nitrogen and oxygen. Their main difference is that nitrates have three oxygen molecules, while nitrites have only two.
Nitrates are so common that they are found naturally in soil, water, and air. They are also incredibly versatile in their compounds.
Nitrates are used in various foods. They can be applied to food coloring and are widely used in the preservation of reconstituted meat and dairy products, and are also used in industrial applications. You can find nitrates in explosives, fertilizers, and even in glass enamels.
What in nitrates and nitrites is dangerous?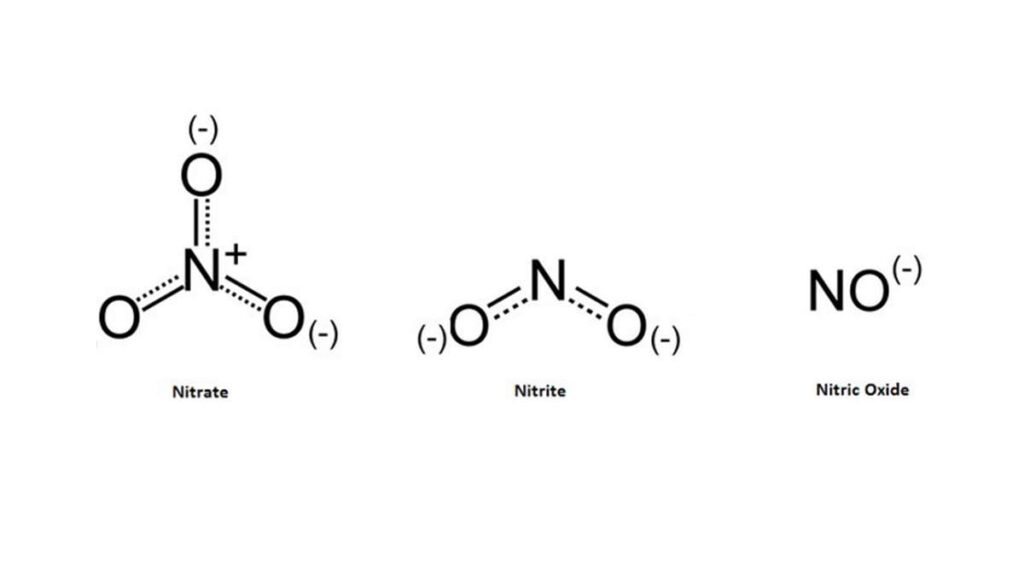
flying reaction. Nitrates don’t pose a big health risk, but when they turn into nitrites, everything changes.
Nitrates are stable compounds, nitrites are not. When nitrates are converted to nitrites, they can be converted to nitric oxide or nitrosamine. Both forms are carcinogenic and have been linked to a range of health problems.
Nitrosamines are formed only in a certain environment, when they are near amino acids and high temperature. Unfortunately, many popular reconstituted meat products are prepared under such conditions.





